What are chlorofluorocarbons?
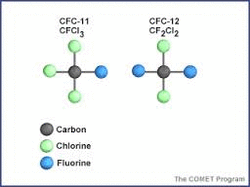
A chlorofluorocarbon (CFC) is a non-toxic molecule that contains the elements carbon, chlorine, and fluorine. They are classified as halocarbons (a class of compounds that contain atoms of carbon and halogen atoms) and are a major source of inorganic chlorine in the stratosphere following their photolytic decomposition by UV radiation. CFCs are used in the manufacture of aerosol sprays, blowing agents for foams and packing materials, as solvents, and as refrigerants. Businesses and consumers use them because they're inexpensive, they don't catch fire easily, and they don't usually poison living things. But the CFCs start eating away at the ozone layer once they get blown into the stratosphere. They can’t be destroyed by rain or broken down in the lower atmosphere. Ozone molecules, which are simply made of three joined oxygen atoms,are always being destroyed and reformed naturally. But CFCs in the air make it very difficult for ozone to reform once it’s broken apart. A relatively small decrease in the stratospheric ozone concentration can result in an increased incidence of skin cancer in humans and genetic damage in many organisms. Ultraviolet radiation in the stratosphere causes the CFC molecules to dissociate, producing chlorine atoms and radicals (i.e., chlorodifluoromethyl radical; free radicals are species that contain one or more unpaired electrons).
